|
6/22/2023 0 Comments Radioactive element definition  
Thus it is the time it takes for exactly half of an arbitrary quantity of that isotope to remain.Īn isotope of a chemical element which is not subject to radioactive decay is described as stable. Radioactive isotopes are those isotopes that are naturally unstable due to the number and configuration of their protons and neutrons. The half-life of a radioactive isotope is the time it takes for exactly half of an arbitrary quantity of that isotope to undergo radioactive decay. However, the new atom may have its own particular (different) mode of decay itself.Īn isotope of a chemical element which is subject to radioactive decay is known as a radioactive isotope.Ī chemical element whose isotopes are all subject to radioactive decay is known as a radioactive element. The meaning of RADIOACTIVITY is the property possessed by some elements (such as uranium) or isotopes (such as carbon 14) of spontaneously emitting energetic particles (such as electrons or alpha particles) by the disintegration of their atomic nuclei also : the rays emitted. half-life, in radioactivity, the interval of time required for one-half of the atomic nuclei of a radioactive sample to decay (change spontaneously into other nuclear species by emitting particles and energy), or, equivalently, the time interval required for the number of disintegrations per second of a radioactive material to decrease by one-half. Having decayed, that atom is now transformed into an atom of a different type, and so is not subject to any further occurrences of that particular decay process. The probability of an individual atom decaying is an intrinsic property of the given chemical element. These pages contain information about the properties of the element, how it moves through the environment, where you may find it (the radiation source) and potential health effects.Radioactive decay is a first-order reaction in which the atoms of a substance may spontaneously break apart, thereby turning into atoms of a different substance. Learn more about radioactive decay.īelow you will find links to basic information about twelve radionuclides encountered in medical, commercial, and military activities. Some radionuclides have half-lives of mere seconds, but others have half-lives of millions of years. Radioactive decay is when a radioisotope transforms into another radioisotope this process emits radiation in some form. It is, in essence, an attribute of individual atomic nuclei. Review the latest IPTEI element-by-element review including a chart of all known stable and radioactive isotopes for each element and examples practical. 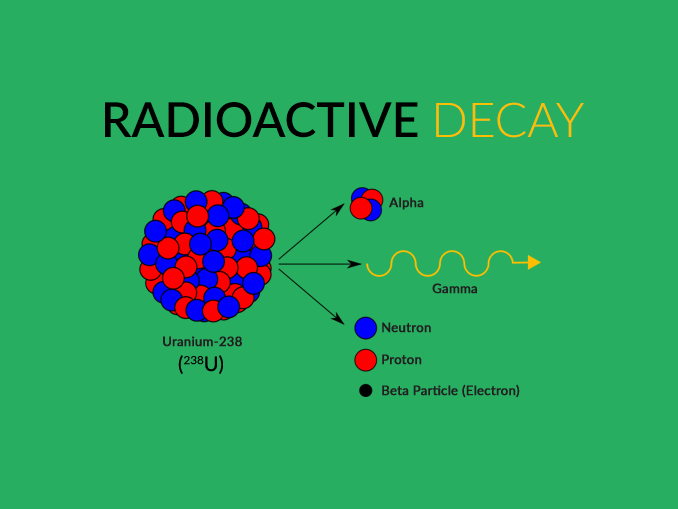
Radioactive half-life is the time required for half of the radioactive atoms present to decay. radioactivity, property exhibited by certain types of matter of emitting energy and subatomic particles spontaneously. Learn the Radiation Basics.Įvery radionuclide emits radiation at its own specific rate, which is measured in terms of half-life. Radioactive (or nuclear) waste is a byproduct from nuclear reactors, fuel processing plants, hospitals and research facilities. Some occur naturally in the environment, while others are man-made, either deliberately or as byproducts of nuclear reactions. Radium-226, Cesium-137, and Strontium-90 are examples of radionuclides. Radioactive forms of elements are called radionuclides radionuclideRadioactive forms of elements are called radionuclides. Radioactivity is defined as the spontaneous emission of radiation from highly energized particles or photons resulting in nuclear reactions. having or producing powerful and dangerous energy that comes from the breaking up of atoms: Uranium is a radioactive material. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
